The effect of the Ln2-P3 motif of the human protein laminin on implant osseointegration

Tissue regeneration using tissue engineering attracted, in recent years, considerable efforts of scientists and manufacturers worldwide. Dental implants, for example, are used after injuries or surgical procedures in order to replace the missing teeth by integrating the implant with the surrounding bone – a process called osseointegration. The majority of dental implants are made of titanium (Ti), and there are modifications on the surface of these implants that might encourage better osseointegration and faster healing [1-4]. Such modifications include coating the implants with bioactive molecules, such as bone morphogenic proteins (BMPs) and calcium phosphate (CaP) [5-7]. Such coatings have been found to improve the healing process and gained clinical success [5,7-11], but problems regarding the stability of the coating for long periods of time still remain [9,11].
Laminin is a protein with cell binding activity [12-15]. Therefore, it is possible that using laminin motifs on the surface of the implant will help improving the bone-implant contact without some of the disadvantages that Cap or BM coatings display [14,16,17].
Recently, peptides have been shown to enhance the functional activity of the scaffold or implant while bypassing or minimizing the immune response of the body [18]. Therefore, they are considered a valuable tool in the field of tissue engineering and provide a promising technique to improve implantation efficiency [19]. In their 2013 article “The effect of the DLTIDDSYWYRI motif of the human laminin a2 chain on implant osseointegration” [20], Kang et al managed to show that the DLTIDDSYWYRI motif of laminin, nicknamed Ln2-P3 can indeed mediate cell attachment [14]. The results of this study are hereby presented.
Ln2-P3 is a major ligand for a protein called syndecan-1 which is known to mediate cell attachment. Therefore, the attachment of osteoblasts-like cells MG63 to plates coated with laminin, Ln2-P3 and scrambled peptide was explored. As expected, both laminin and Ln2-P3 strongly promoted the attachment and spreading of the cells (Figure 1), compared to the controls BSA and scrambled peptide (SP). In fact, LN2-P3, which is, as mentioned above, one of the motifs comprising laminin, and the full-length laminin had the same effect on cell attachment.
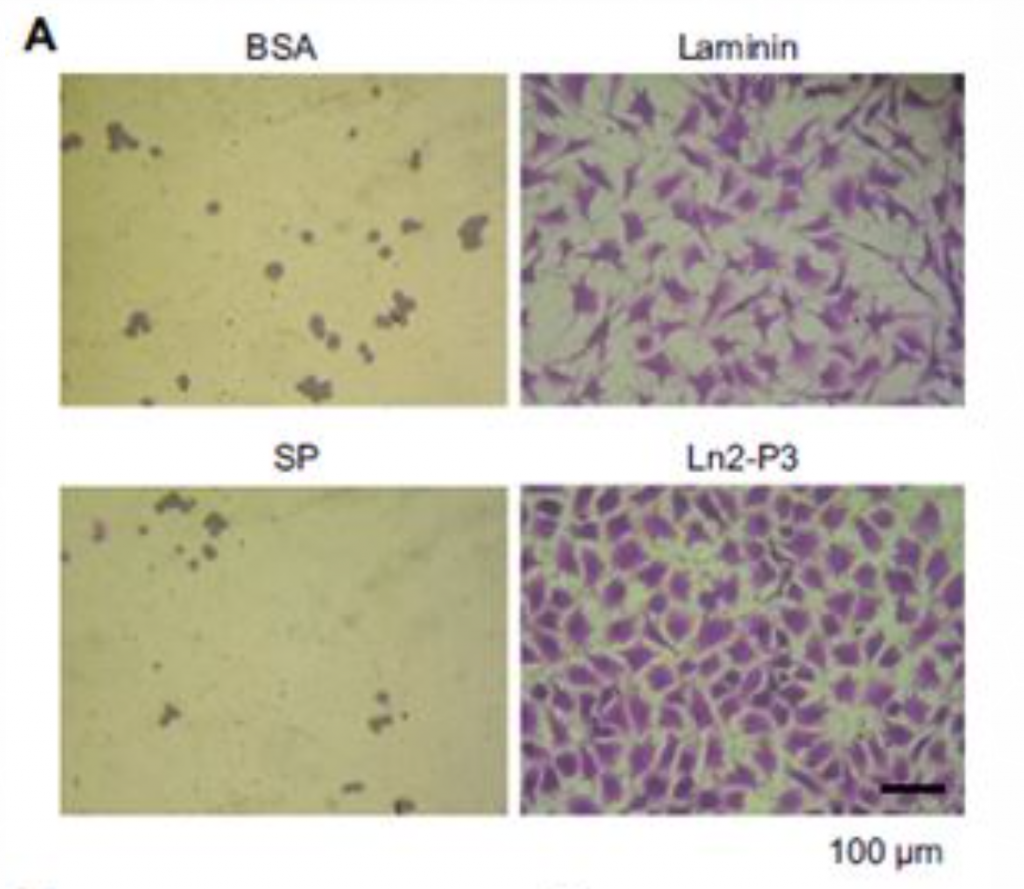
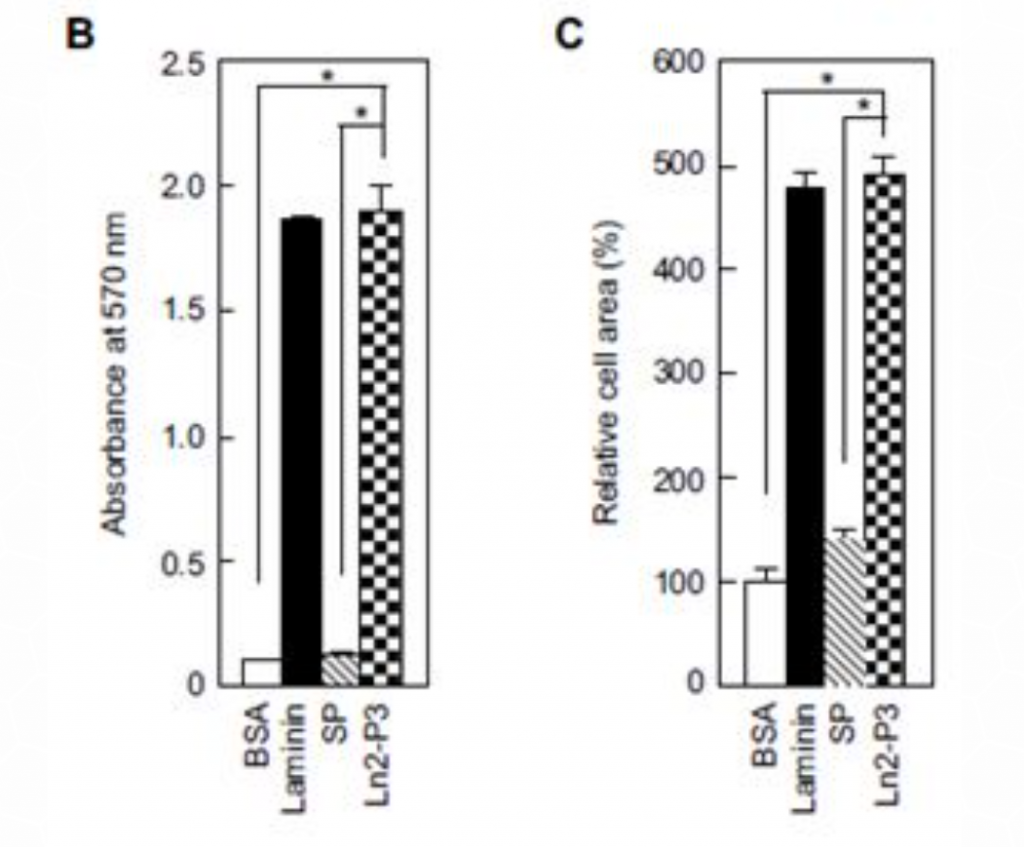
Figure 1: Attachment and spreading of osteoblast-like MG63 cells seeded onto plates coated with laminin, Ln2-P3, BSA and scrambled peptide (SP). (A) MG63 cells adhering to the culture plates coated with laminin (1.3 mg/cm2), Ln2-P3 (21 mg/cm2), BSA (1%) or SP (21 mg/cm2) for 3 hours. (B) Quantification of the attachment of MG63 cells which were seeded on plates coated with laminin, Ln2-P3, BSA or SP for 1 hour. (C) Quantification of the spreading of MG63 cells induced by laminin, Ln2-P3, BSA or SP. Cell area was measured by using
Image-Pro Plus software. Data are presented as the percentage of the value for cells seeded on BSA-coated plates. Results are resented as the mean ± SD (N=4).
ANOVA: p < 0.05. Pairwise comparisons: *p < 0.01.
Osteogenesis markers can be generally divided into three groups: early, intermediate and late. The effect of Ln2-P3 on early/intermediate markers (type I collagen) and intermediate/late markers (Cbfa-1, bone sialoprotein and osteocalcin) was evaluated by using MG63 cells cultured for 1 or 7 days on different discs (Figure 2). When the cells were cultured on Ti discs treated with Ln2-P3 for 7 days, a significant increase in mRNA levels of all aforementioned factors was observed. These results indicate that Ln2-P3 has an osteogenic effect on the osteoblasts-like cell line MG63.
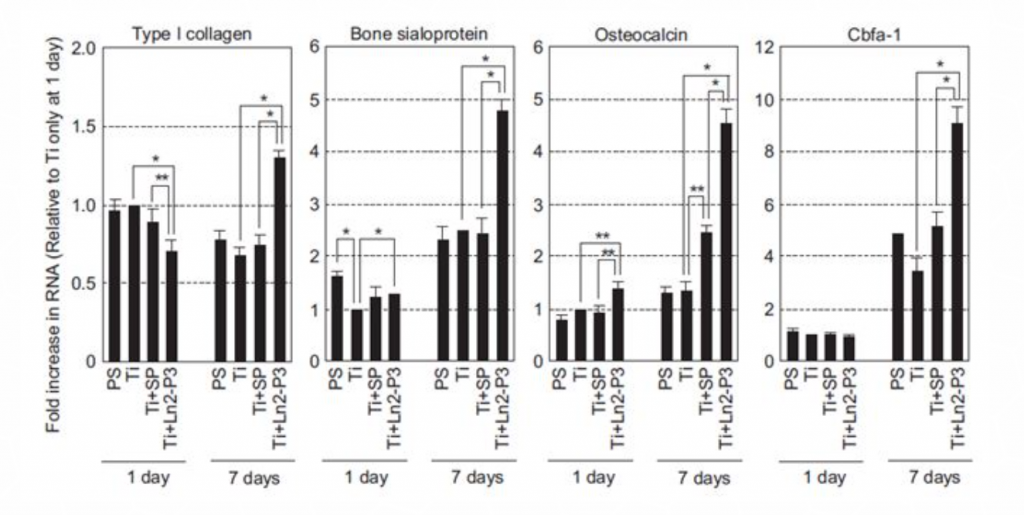
Figure 5. mRNA levels of osteogenic markers in MG63 osteoblast-like cells grown on different materials. MG63 cells were seeded onto polystyrene (PS) or Ti discs coated with scrambled peptide (SP) or Ln2-P3 (21 mg/cm2), and cultured on the discs for 1 day and 7 days. The expression of osteogenic marker genes – type I collagen, bone sialoprotein, osteocalcin, and Cbfa-1 – was determined by real-time RT-PCR. RT. Results presented as mean ± SD (N=3). ANOVA: p < 0.05. Pairwise comparisons: *p < 0.01; **p < 0.05.
Another major factor that serves as an early/intermediate marker for osteogenesis is alkaline phosphatase (ALP). ALP is also used to indicate osteoblasts differentiation, as increased ALP activity is associated with higher differentiation levels. ALP activity was measured in MG63 cells cultured on polystyrene or treated Ti discs for 1 and 3 days (Figure 3). The results show increased ALP activity after both time points in cells grown on Ti discs coated with Ln2-P3 compared to cells grown on untreated Ti discs.
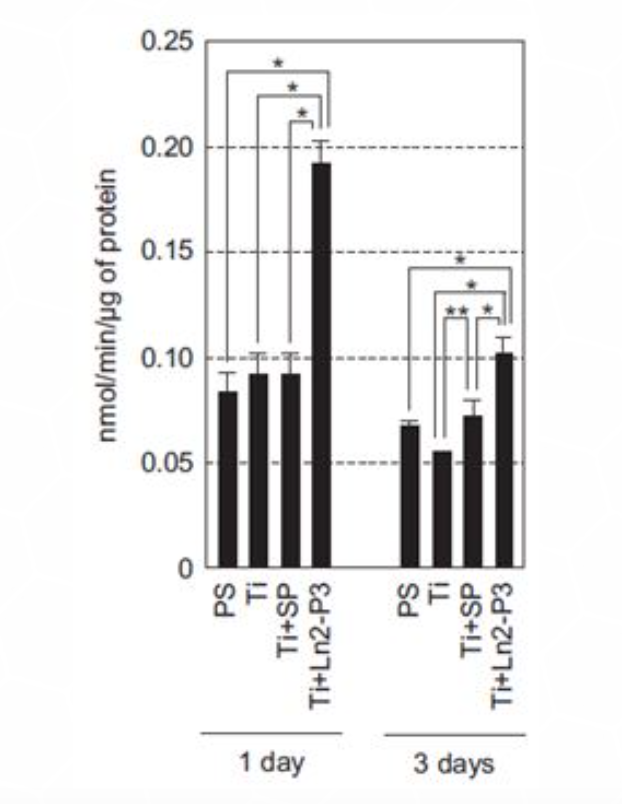
Figure 6: Alkaline phosphatase (ALP) activity of MG63 cells grown on different materials. The culture conditions were the MG63 cells were seeded onto polystyrene (PS) or Ti discs coated with scrambled peptide (SP) or Ln2-P3 (21 mg/cm2), and cultured on the discs for 1 day and 3 days. ALP expression was determined by real-time RT-PCR. Results presented as mean ± SD (N=3). ANOVA: p < 0.05. Pairwise comparisons: *p < 0.01; **p < 0.05.
The results show that Ln2-P3, a motif of the protein laminin, encourage cell attachment and spreading, which are qualities that contribute to the success of dental implants and might indicate that implants coated with Ln2-P3 will have higher success rates that un-coated implants. Higher expression of osteogenic marker genes and increased ALP activity in MG63 cells grown on Ln2-P3-coated Ti surfaces also indicate that Ln2-P3 can improve osteoblasts growth and differentiation – an essential stage in bone healing around the implant.
Bibliography
[1] Cooper LF. A role for surface topography in creating and maintaining bone at titanium endosseous implants. J Prosthet Dent 2000;84:522-34.
[2] Ellingsen JE. Surface configurations of dental implants. Periodontol 2000 1998;17:36-46.
[3] Ellingsen JE, Johansson CB, Wennerberg A, Holmen A. Improved retention and bone-tolmplant contact with fluoride-modified titanium implants. Int J OralMaxillofac Implants 2004;19:659-66.
[4] Wennerberg A, Albrektsson T, Andersson B, Krol JJ. A histomorphometric and removal torque study of screw-shaped titanium implants with three different surface topographies. Clin Oral Implants Res 1995;6:24-30.
[5] Harada Y, Wang JT, Doppalapudi VA, Willis AA, Jasty M, Harris WH, et al. Differential effects of different forms of hydroxyapatite and hydroxyapatite/ tricalcium phosphate particulates on human monocyte/macrophages in vitro. J Biomed Mater Res 1996;31:19-26.
[6] Lee DW, Yun YP, Park K, Kim SE. Gentamicin and bone morphogenic protein-2 (BMP-2)-delivering heparinized-titanium implant with enhanced antibacterial activity and osteointegration. Bone 2012;50:974-82.
[7] Lee JJ, Rouhfar L, Beirne OR. Survival of hydroxyapatite-coated implants: a meta-analytic review. J Oral Maxillofac Surg 2000;58:1372;9 [discussion 1379-80].
[8] Rungcharassaeng K, Lozada JL, Kan JY, Kim JS, CampagniWV, Munoz CA. Periimplant tissue response of immediately loaded, threaded, HA-coated implants: 1-year results. J Prosthet Dent 2002;87:173-81.
[9] Sun L, Berndt CC, Gross KA, Kucuk A. Material fundamentals and clinical performance of plasma-sprayed hydroxyapatite coatings: a review. J Biomed Mater Res 2001;58:570-92.
[10] Yeo IS, Han JS, Yang JH. Biomechanical and histomorphometric study of dental implants with different surface characteristics. J Biomed Mater Res B Appl Biomater 2008;87:303-11.
[11] Kang JD. Another complication associated with rhBMP-2? Spine J 2011;11:517e9.
[12] Suzuki N, Yokoyama F, Nomizu M. Functional sites in the laminin alpha chains. Connect Tissue Res 2005;46:142-52.
[13] Vuolteenaho R, Nissinen M, Sainio K, Byers M, Eddy R, Hirvonen H, et al. Human laminin M chain (merosin): complete primary structure, chromosomal assignment, and expression of the M and A chain in human fetal tissues. J Cell Biol 1994;124:381-94.
[14] Jung SY, Kim JM, Kang HK, Jang da H, Min BM. A biologically active sequence of the laminin alpha2 large globular 1 domain promotes cell adhesion through syndecan-1 by inducing phosphorylation and membrane localization of protein kinase Cdelta. J Biol Chem 2009;284:31764-75.
[15] Jung SY, Kim JM, Min SK, Kim OB, Jang da H, Kang HK, et al. The potential of laminin-2-biomimetic short peptide to promote cell adhesion, spreading and migration by inducing membrane recruitment and phosphorylation of PKC delta. Biomaterials 2012;33:3967-79.
[16] Hoffman MP, Nomizu M, Roque E, Lee S, Jung DW, Yamada Y, et al. Laminin-1 and laminin-2 G-domain synthetic peptides bind syndecan-1 and are involved in acinar formation of a human submandibular gland cell line. J Biol Chem 1998;273:28633-41.
[17] Nomizu M, Song SY, Kuratomi Y, Tanaka M, Kim WH, Kleinman HK, et al. Active peptides from the carboxyl-terminal globular domain of laminin alpha2 and Drosophila alpha chains. FEBS Lett 1996;396:37-42.
[18] Berke Z, Palmer S, Bergman T, Wester D, Svedmyr J, Linder S, et al. A short peptide eluted from the H-2Kb molecule of a polyomavirus-positive tumor corresponds to polyomavirus large T antigen peptide at amino acids 578 to 585 and induces polyomavirus-specific immunity. J Virol 1996;70: 3093-7.
[19] Min SK, Lee SC, Hong SD, Chung CP, Park WH, Min BM. The effect of a laminin-5-derived peptide coated onto chitin microfibers on re-epithelialization in early-stage wound healing. Biomaterials 2010;31:4725-30.
[20] Kang HK, Kim OB, Min SK, Jung SY, Jang da H, Kwon TK, Min BM, Yeo IS. The effect of the DLTIDDSYWYRI motif of the human laminin α2 chain on implant osseointegration. Biomaterials 2013;3416:4027-37




